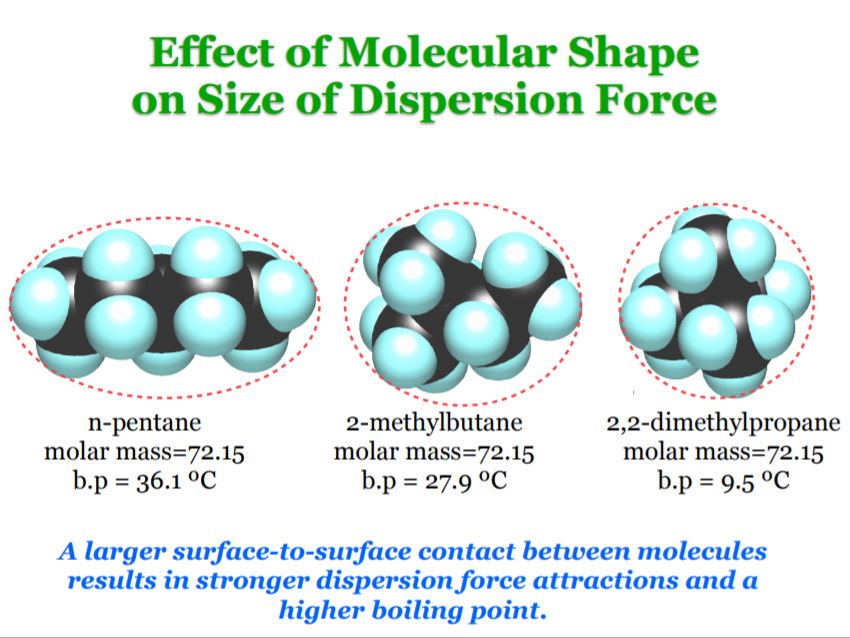
When considering a substance, following the steps below will help you determine what type(s) of intermolecular forces exist in the substance.Ĭlick on each number to see steps to follow. I learned that what results in London fores between molecules is the 'cloud of electrons' that results in an instantaneous dipole and induced dipole, which eventually results into. With these overlapping ranges, how can the intermolecular forces of two different substances be compared? The summary below will answer this question. 'London (dispersion) forces are responsible for the fact that non-polar substances can be condensed to form liquids and sometimes solids at low temperatures'. 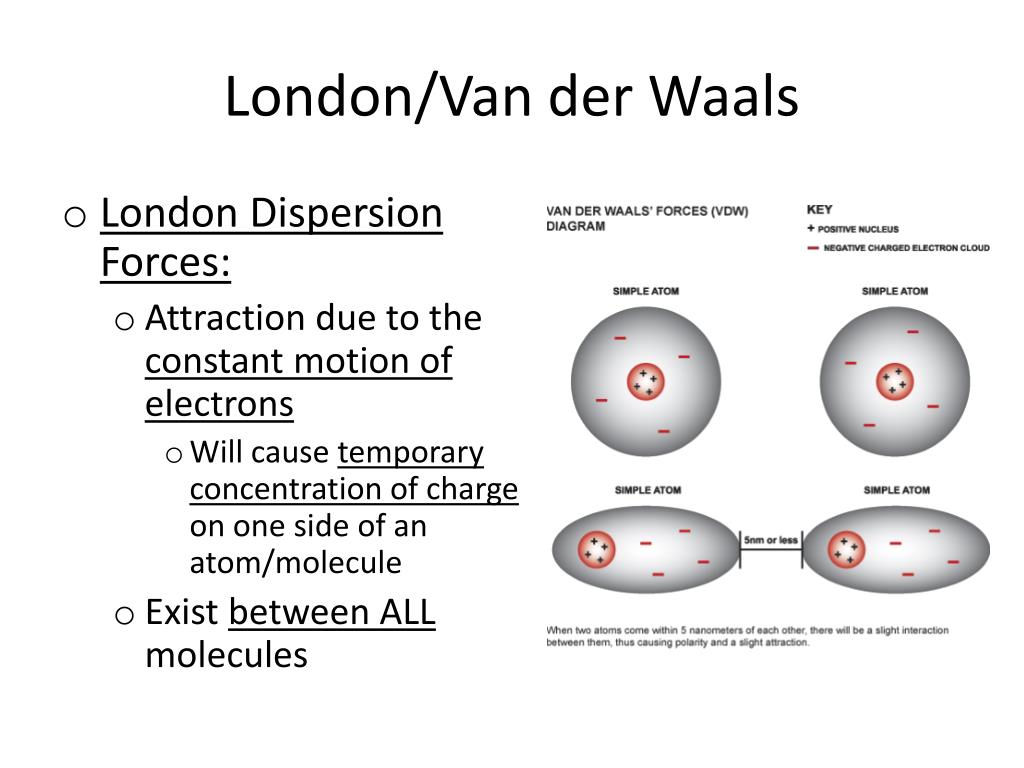
This means a statement such as "Dipole-dipole forces are stronger than London forces" can not be made. fluctuations in molecules (known as London forces, or dispersion forces) are present even between permanently polar molecules and produce, generally. In this article we will cover Dispersion Forces or London Forces: Definition, Examples, Types and Formula, Symmetrical, No dipole. More importantly, the ranges for the various intermolecular forces overlap. London dispersion force is the weakest type of intermolecular force caused by the uneven distribution of electrons in molecules. * For comparison, the approximate magnitude of covalent bonds is 100-400 kJ/mol.Īs you can see in the table, the strength of each type of intermolecular force covers a range of values. The table below compares the strength of various intermoelcular forces. While intermolecular forces are strong enough to keep molecules together in the solid and liquid state, they are not nearly as strong as covalent bonds.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
